(All Science Awareness factsheets displayed on the menu at the end of the page)
Antibiotic Multiresistant Gram-Negative Bacteria
THE PROBLEM:
The rapid spread of antibiotic resistant bacteria is one of the biggest threats to global health, with international organizations such as the World Health Organization and the European Centre for Disease Prevention and Control, using terms such as “crisis”, “catastrophic consequences” and “nightmare scenario” [1, 2].
Most classes of antibiotics in current use were discovered more than forty years ago; since then, most new approved drugs are based on chemical modifications of existing scaffolds [3]. Today there is an urgent need to define new approaches for tackling multidrug resistant pathogens, with only a limited number of large pharma companies still active in the area [4]. Some international initiatives have been launched in the last few years, but limited efforts are being focused on early discovery programmes [5, 6].
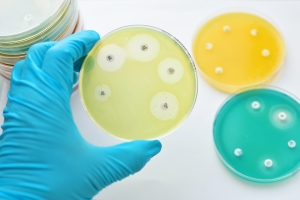
Antimicrobial susceptibility testing in petri dish
The ENDOBIOS Antibiotics Discovery Programme
THE “ENCRYPTED” SOLUTION:
Microorganisms, particularly fungi and bacterial actinomycetes [7], have traditionally been one of the most important sources of drugs. Of all antibiotics used in clinics today, 90% are derived from microorganisms [8].
In contrast to the unfavourable antibiotic discovery environment, studies of microbial genomes have revealed an overwhelming biosynthetic potential for the production of new natural compounds [9-11]. The majority of these microbial gene clusters are not expressed under normal laboratory conditions, which represents a treasure trove for the discovery of new bioactive natural compounds [12]. One recent estimate place the number of discovered antibiotics as only 1% from all microbes [13].
THE DISRUPTIVE ENDOBIOS-“ENDOBAC” TECHNOLOGY:
Current methods for screening new bioactive natural compounds, ranging from classic random approaches to metabolic engineering are costly, labour-intensive, and ineffective [14, 15], failing to explore the overwhelming potential of cryptic microbial natural compounds.
The ENDOBIOS® – “ENDOBAC” disruptive technology was developed after the discovery of compounds that are perceived by environmental fungi and bacteria as a bacterial chemical attack. Their response is the induced production of cryptic antibiotics, that are silent when cultivating said environmental fungi and bacteria in standard laboratory cultures. These responses are hypothesized to be part of the chemical microbial “immune system”, optimised for millions of years.

An environmental fungus in a petri dish. Upon cultivation with proprietary inducers of cryptic antibiotics, fungal extracts are tested against clinical bacteria.
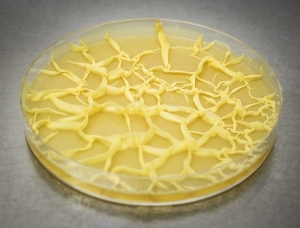
The bacterium Massilia timonae in a biofilm-like culture
The ENDOBAC concept was generated after merging the two ENDOBIOS® areas of expertise: 15+ years in bioactive natural products from microbes, with 45+ new natural compounds characterized (e.g. [16-22], and 10+ years in the under-explored field of endosymbiotic bacteria in eukaryotic hosts [23]. The intellectual property supporting our methodology includes multidisciplinary layers (e.g. unpublished features in endosymbiotic biology), aims to solve the lack of antibiotics with the discovery of new chemical scaffolds and will be strategically applied against the most problematic gram-negative clinical pathogens.
References
- Martin-Loeches, I., et al., J Infect, 2015, 70(3): p. 213-22.
- WHO. Antibiotic resistance. 2017 November 2017 March 2018]; Available from: http://www.who.int/mediacentre/factsheets/antibiotic-resistance/en/.
- Genilloud, O.. Nat Prod Rep, 2017. 34(10): p. 1203-1232.
- Page, M.G. and K. Bush, Curr Opin Pharmacol, 2014. 18: p. 91-7.
- Rex, J.H., Nat Rev Microbiol, 2014. 12: p. 231.
- Cooper, M.A., Nat Rev Drug Discov, 2015. 14(9): p. 587-8.
- Zida, A., et al., J Mycol Med, 2017. 27(1): p. 1-19.
- Wohlleben, W., et al., Microb biotechnol, 2016. 9(5): p. 541-548.
- Hopwood, D., American Society for Microbiology, 2008.
- Cimermancic, P., et al., Cell, 2014. 158(2): p. 412-421.
- Katz, L. and R.H. Baltz, J Ind Microbiol Biotechnol, 2016. 43(2-3): p. 155-76.
- Okada, B.K. and M.R. Seyedsayamdost, FEMS Microbiol Rev, 2017. 41(1): p. 19-33.
- Watve, M.G., et al., Arch Microbiol, 2001. 176(5): p. 386-90.
- Lewis, K., Nat Rev Drug Discov, 2013. 12(5): p. 371-87.
- Ochi, K., J Antib, 2016. 70: p. 25.
- Almeida, C., et al., J Nat Prod, 2011. 74(1): p. 21-5.
- Almeida, C., et al., Beilstein J Org Chem, 2011. 7: p. 1636-42.
- Almeida, C., et al., Chemistry, 2012. 18(28): p. 8827-34.
- Almeida, C., et al., J Nat Prod, 2013. 76(3): p. 322-6.
- Almeida, C., et al., J Antibiot (Tokyo), 2014. 67(5): p. 421-3.
- Almeida, C., et al., Org Lett, 2016. 18(3): p. 528-31.
- Almeida, C., et al., J Nat Prod, 2018. 81(6): p. 1488-1492.
- Almeida, C., et al., Appl Environ Microbiol, 2018. 84, 15, e00660-18.
